
Vibrations of the H2O Molecule
Source: "Water Absorption Spectrum"
(in cache)
in "Water Structure and Science",
by Martin Chaplin

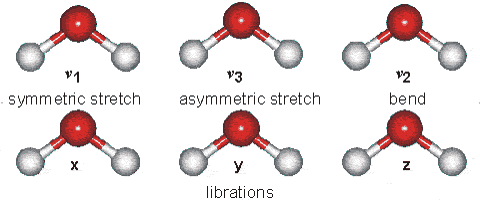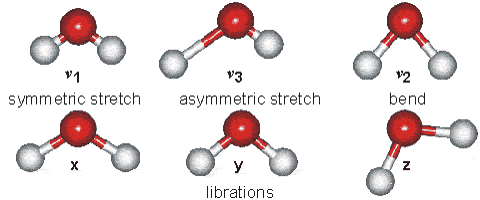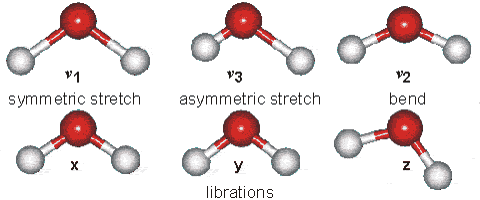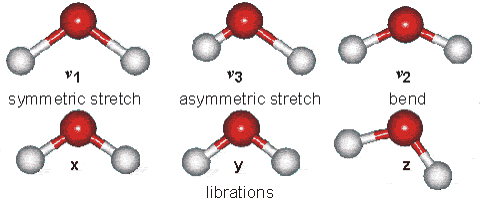

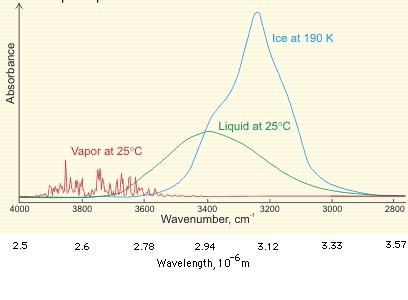
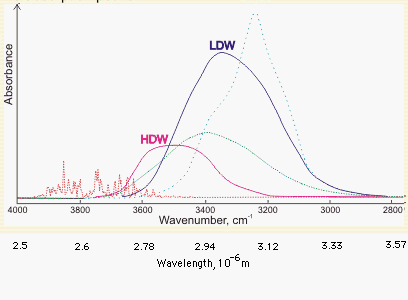
Above is shown a comparison of the gas, liquid and solid spectra of the same amount of H2O [1392]. On the right half of the Figure, the high (HDL) and low (LDL) density liquid water forms are shown [1738].
As seen, increased strength of hydrogen bonding typically shifts the stretch vibration to lower frequencies (red-shift) with greatly increased intensity in the infrared (but not Raman) due to the increased dipoles. Blue-shifting hydrogen bonds are described elsewhere.
8. D. Eisenberg and W. Kauzmann, The structure and properties of water (Oxford University Press, London, 1969); (b) The dodecahedral interstitial model is described in L. Pauling, The structure of water, In Hydrogen bonding, Ed. D. Hadzi and H. W. Thompson (Pergamon Press Ltd, London, 1959) pp. 1-6
177. H. R. Zelsmann, Temperature dependence of the optical constants for liquid H2O and D2O in the far IR region, J. Mol. Struct. 350 (1995) 95-114.
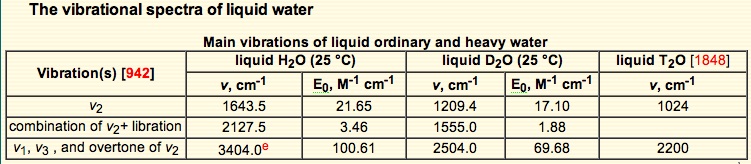
942. J.-J. Max and C. Chapados, Isotope effects in liquid water by infrared spectroscopy. III. H2O and D2O spectra from 6000 to 0 cm-1, J. Chem. Phys. 131 (2009) 184505. see earlier papers in [1502].
1392. G. E. Ewing, M. Foster, W. Cantrell and V. Sadtchenko, Thin film water on insulator surfaces, in Water in Confining Geometries, eds V. Buch and J. P. Devlin, (Springer-Verlag, Berlin, 2003) pp. 179-211. The original figure was from David Weis.
1502. P. Larouche, J.-J. Max and C. Chapados, Isotope effects in liquid water by infrared spectroscopy. II. factor analysis of the temperature effect on H2O and D2O, J. Chem. Phys. 129 (2008) 064503. The conclusions in this work was shown to be compatible with the works by Y. Maréchal, Infrared spectra of water. I. Effect of temperature and of H/D isotopic dilution J. Chem. Phys. 95 (1991) 5565-5573, and Zelsmann [177].
1738. Y. Maréchal, The molecular structure of liquid water delivered by absorption spectroscopy in the whole IR region completed with thermodynamics data, J. Mol. Structure 1004 (2011) 146-155.
Version: 23. December 2012
Address of this page
Joachim Gruber
-Noncommercial-No Derivative Works 2.0 UK: England & Wales License